 |
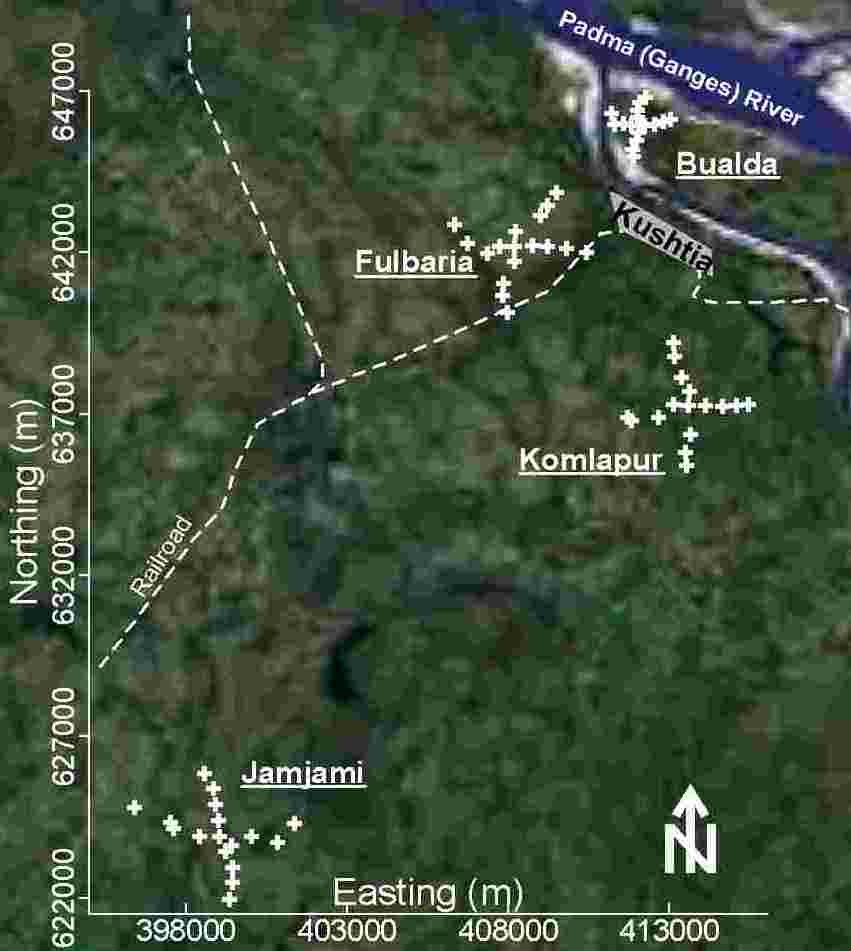
A total of 71 samples were collected from 67 randomly selected tubewells in these 4 neighborhoods. A total of 18 samples were collected from 17 tubewells in each of 3 neighborhoods (Bualda, Fulbaria, and Jamjami). Access was denied at 1 sampling location; therefore, a total of 17 samples were collected from 16 tubewells in the fourth neighborhood (Komlapur). To the extent possible, the sampled tubewells in each neighborhood were distributed at 500-meter intervals along perpendicular axes that radiated in 4 equal lengths from the center (Figure 1). Two samples were collected from the centermost tubewell in each neighborhood. The results for each analyte from each of these 4 centermost tubewells were averaged. One sample was collected from each of the remaining tubewells. The northings and eastings of these tubewells were measured using a Global Positioning System 12 Channel Personal Navigator™ (Garmin International, Olathe, KS, USA).
Established collection, preservation, and storage methodologies were used to ensure that each sample was representative of groundwater quality (APHA et al. 2005; Frisbie et al. 2005). Accordingly, all sampled tubewells were purged by pumping vigorously for 10 minutes immediately before sample collection. All samples were collected directly into polyethylene bottles. These samples were not filtered. Samples were analyzed immediately after collection with pH paper, preserved by acidification to pH < 2 with 5.0 Molar (M) hydrochloric acid (HCl; BDH Laboratory Supplies, product number 101256J, Poole, England), and stored in ice-packed coolers. The temperature of all stored samples was maintained at 0º to 4º Celsius (C) until immediately before analysis at laboratories in Dubai, France, and Vermont.
Samples were shipped to Dubai and analyzed for As by the arsenomolybdate method (Frisbie et al. 2005). After which, the samples were shipped to France and analyzed for Ba, Cr, Mn, Mo, Ni, Pb, Se, U, and Zn by inductively coupled plasma mass spectrometry (ICPMS; PlasmaQuad PQ2+ Spectrometer, Fisons/VG Analytical, Manchester, UK; APHA et al. 2005). Finally, the samples were shipped to Vermont and analyzed for B by the azomethine H method, Fe by flame atomic absorption spectroscopy (FAAS; 210VGP Atomic Absorption Spectrometer, Buck Scientific, East Norwalk, CT, USA; APHA et al. 2005), and Sb by graphite furnace atomic absorption spectroscopy (GFAAS; 210VGP Atomic Absorption Spectrometer, Buck Scientific; APHA et al. 2005).