| Element | Average Concentration (µg/L) |
WHO Health-Based Guideline (µg/L) |
% of Unsafe Tubewells a |
| As B Ba Cr Fe Mn Mo Ni Pb Sb Se c U Zn c |
29 < 50 140 4.7 2,700 800 1.4 11 0.5 1.6 < 1 2.5 15 |
10 500 700 50 NA b 400 70 70 10 20 10 2 NA |
33 0 0 1 NA 78 0 1 1 0 0 48 NA |
| As | B | Ba | Cr | Fe | Mn | Mo | Ni | Pb | Sb | Se | U | Zn | pH | Depth | Age | Users | |
| As | 1.00a | ||||||||||||||||
| B | 0.81a | 1.00a | |||||||||||||||
| Ba | 0.26b | 0.40a | 1.00a | ||||||||||||||
| Cr | 0.82a | 0.92a | 0.30b | 1.00a | |||||||||||||
| Fe | 0.82a | 0.92a | 0.40a | 0.97a | 1.00a | ||||||||||||
| Mn | 0.46a | 0.31b | 0.19 | 0.26b | 0.21 | 1.00a | |||||||||||
| Mo | 0.28b | 0.05 | 0.16 | -0.03 | -0.01 | 0.28b | 1.00a | ||||||||||
| Ni | 0.13 | 0.07 | 0.07 | 0.10 | 0.09 | -0.09 | -0.05 | 1.00a | |||||||||
| Pb | 0.83a | 0.94a | 0.33a | 0.98a | 0.96a | 0.28b | -0.02 | 0.09 | 1.00a | ||||||||
| Sb | 0.34a | 0.33a | 0.56a | 0.24 | 0.31b | 0.38a | 0.29b | 0.40a | 0.28b | 1.00a | |||||||
| Se | 0.32a | 0.31b | 0.48a | 0.30b | 0.32a | 0.31b | 0.10 | 0.11 | 0.35a | 0.30b | 1.00a | ||||||
| U | -0.02 | 0.07 | -0.27b | 0.04 | -0.05 | 0.18 | -0.21 | -0.02 | 0.08 | 0.04 | 0.09 | 1.00a | |||||
| Zn | 0.67a | 0.69a | 0.29b | 0.73a | 0.74a | 0.15 | 0.19 | 0.03 | 0.70a | 0.21 | 0.29b | -0.14 | 1.00a | ||||
| pH | -0.02 | -0.01 | -0.11 | 0.02 | 0.00 | 0.05 | -0.10 | -0.20 | 0.03 | -0.24 | 0.07 | 0.07 | 0.05 | 1.00a | |||
| Depth | -0.10 | -0.07 | -0.17 | 0.00 | -0.06 | 0.06 | -0.15 | -0.22 | 0.01 | -0.28b | -0.19 | 0.18 | 0.01 | 0.10 | 1.00a | ||
| Age | -0.08 | -0.03 | -0.03 | -0.05 | -0.02 | 0.02 | -0.17 | -0.11 | -0.03 | 0.01 | 0.16 | 0.27b | -0.17 | 0.11 | -0.13 | 1.00a | |
| Users | -0.07 | -0.06 | -0.11 | -0.05 | -0.06 | -0.08 | -0.07 | -0.03 | -0.04 | -0.04 | 0.05 | 0.16 | -0.12 | 0.07 | 0.02 | 0.10 | 1.00a |
The first study on humans of the effects of chronic U ingestion from drinking water showed adverse kidney function with the proximal tubule as the site of toxicity (Zamora et al. 1998). Later, a much larger study on exposure of humans to U in drinking water revealed nephrotoxic effects even at low concentrations without a clear threshold (Kurittio et al. 2002). In another study, the same authors found that people who drank water with elevated concentrations of U had indications that, in addition to kidneys, bone may be another target of toxicity (Kurittio et al. 2005).
Antimony trioxide (Sb2O3) is "possibly carcinogenic to humans" by inhalation exposure. In contrast, the effect of Sb in drinking water on cancer in humans has not been adequately studied. Antimony in drinking water did not increase the incidence of tumors in laboratory mice and rats (WHO 1996a; WHO 1996b; WHO 2004; WHO 2006). The WHO guideline for Sb in drinking water was calculated using the NOAEL for decreased water intake, food intake, and body weight in laboratory rats (WHO 2004; WHO 2006).
Selenium prevents the cytotoxic effects of As (Biswas et al. 1999). Unfortunately, the food crops in Bangladesh are sometimes deficient in Se (Ortega et al. 2003), and the drinking water in Bangladesh is often deficient in Se (Frisbie et al. 2002). Therefore, it is possible that this lack of Se in food and drinking water might cause a magnification of As toxicity.
In Bangladesh, the severity of chronic As poisoning may be magnified by a lack of dietary Zn (Frisbie et al. 2002; Ortega et al. 2003). Zinc promotes the repair of tissues damaged by As (Engel et al. 1994). Food, not drinking water, is the major source of dietary Zn (WHO 1996a), but the agricultural soils, food crops, and diet in Bangladesh are often deficient of Zn (Brammer 1996; Ortega et al. 2003). Therefore, it is possible that this lack of Zn in soils, food, and drinking water may cause a magnification of As toxicity.
| Element | Average Concentration (µg/L) |
WHO Health-Based Guideline (µg/L) |
% of Unsafe Tubewells a |
| As B Ba Cr Fe Mn Mo Ni Pb Sb Se c U Zn c |
84 <50 220 9.5 7,300 870 2.0 31 1.2 2.3 <1 0.9 21 |
10 500 700 50 NA b 400 70 70 10 20 10 2 NA |
100 0 0 5 NA 59 0 5 5 0 0 14 NA |
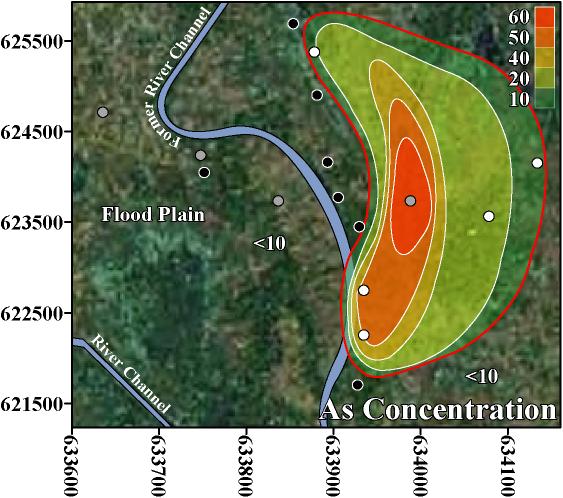
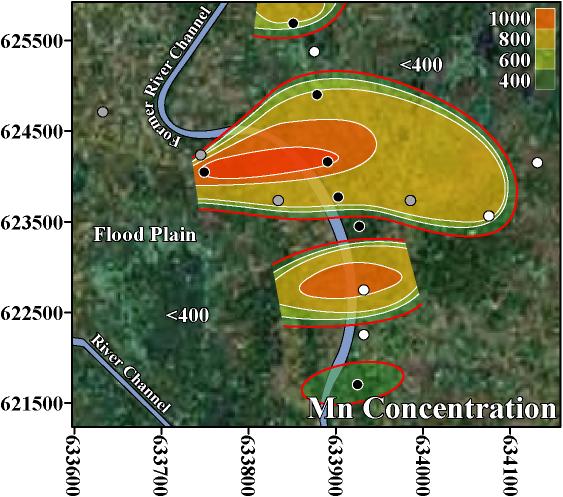
In this neighborhood-scale study and in 2 national-scale studies of Bangladesh, As, Mn, U, Pb, Ni, Cr, B, Ba, and Mo were found above WHO health-based drinking water guidelines (Table 1; BGS/DPHE 2001; Frisbie et al. 2002). In Bualda as the concentration of As increases there are statistically significant increases in the concentrations of Mn, Pb, Ni, Cr, and B (Table 4). In Jamjami as the concentration of As increases there are statistically significant increases in the concentrations of Pb, Ni, and Ba (Table 4). In Komlapur as the concentration of As increases there are statistically significant increases in the concentrations of Cr, and Ba (Table 4). Finally, in the entire region as the concentration of As increases there are statistically significant increases in the concentrations of Mn, Pb, Cr, B, Ba, and Mo (Table 2).
| Element | As | |||
| Bualda | Fulbaria | Jamjami | Komlapur | |
| As | 1.00a | 1.00a | 1.00a | 1.00a |
| B | 0.91a | 0.18 | -0.03 | -0.19 |
| Ba | 0.16 | 0.14 | 0.69a | 0.74a |
| Cr | 0.91a | 0.23 | 0.45 | 0.60b |
| Fe | 0.91a | 0.21 | 0.61a | 0.66a |
| Mn | 0.49b | 0.33 | -0.04 | -0.39 |
| Mo | 0.21 | 0.09 | -0.24 | 0.27 |
| Ni | 0.91a | 0.25 | 0.49b | 0.30 |
| Pb | 0.91a | 0.20 | 0.52b | 0.24 |
| Sb | 0.37 | -0.18 | 0.39 | 0.26 |
| Se | 0.40 | 0.14 | 0.47 | 0.53b |
| U | 0.03 | -0.16 | -0.55b | -0.30 |
| Zn | 0.96a | -0.14 | 0.34 | 0.06 |
| pH | 0.27 | -0.32 | 0.09 | 0.08 |
| Depth | 0.07 | -0.09 | -0.69a | -0.03 |
| Age | -0.19 | 0.03 | -0.34 | 0.01 |
| Users | -0.23 | -0.17 | -0.33 | -0.26 |
Almost all of the home-scale drinking water treatment systems currently being used in Bangladesh have been designed to remove As, but not these other toxic elements. The statistically significant increases in toxic elements with As suggest that these treatment systems should be further evaluated for the removal of Mn, Pb, Ni, Cr, B, Ba, Mo, and possibly other elements.
| Element | Average Concentration (µg/L) |
WHO Health-Based Guideline (µg/L) |
% of Unsafe Tubewells a |
| As B Ba Cr Fe Mn Mo Ni Pb Sb Se c U Zn c |
<7 <50 110 2.4 400 770 1.2 1.0 <0.2 1.2 <1 3.2 12 |
10 500 700 50 NA b 400 70 70 10 20 10 2 NA |
0 0 0 0 NA 87 0 0 0 0 0 64 NA |
In response to this finding that Mn, U, and possibly other toxic elements commonly occur at unsafe concentrations even when As is at safe concentrations, the following 3-step testing program is proposed to provide safe drinking water in western Bangladesh, and possibly the entire country. This testing program is economical because it prioritizes the analysis of toxic elements, and stops these analyses as soon as a sample is found to be unsafe for use as drinking water.
First, the toxicity and distribution of As relative to Mn, U, Pb, Ni, Cr, B, Ba, and Mo suggest that the current practice of sampling and testing every tubewell in Bangladesh for As to find the safest sources of drinking water stay as the highest public health priority. Arsenic is expected to cause at least 150,000 extra cancer deaths during the life spans of the current population of Bangladesh (Frisbie et al. 2005). In contrast, the risk to public health in Bangladesh is smaller for Mn, U, Pb, Ni, Cr, B, Ba, and Mo than for As (Frisbie et al. 2002; WHO 1996b; WHO 1998b). Under conditions of limited resources, testing of these toxic elements must be prioritized.
Second, the high concentrations of As, Mn, and U relative to Pb, Ni, Cr, B, Ba, and Mo suggest that if a sample meets the WHO guideline for As, it should be retested for Mn and U. This will identify tubewells with safe concentrations of As, Mn, and U for additional evaluation as a potential drinking water supply in these neighborhoods without the cost or delay of testing for all 9 elements. For example, 1 tubewell in Fulbaria, 1 tubewell in Jamjami, and 1 tubewell in Komlapur did not exceed WHO health-based drinking water guidelines for As, Mn, and U.
Third, if a sample meets the WHO guidelines for As, Mn and U, then it should be retested for Pb, Ni, Cr, B, Ba, and Mo. All tubewells that do not exceed WHO guidelines for these 9 elements could be used as public drinking water supplies. For example, the same 1 tubewell in Fulbaria, 1 tubewell in Jamjami, and 1 tubewell in Komlapur did not exceed any WHO health-based drinking water guidelines and could supply safe drinking water to the residents of each neighborhood.
Testing for just As and then asking the owners of safe tubewells to share drinking water with their less fortunate neighbors has been a highly successful public health strategy in Bangladesh. Over 90% of western Bangladeshis share drinking water (Frisbie et al. 2005). The 3-step testing program builds on this success by testing for all known toxic elements in Bangladesh’s drinking water, not just As.
Unfortunately, no tubewells in Bualda met WHO guidelines for all elements; therefore, drinking water treatment will likely be required in this neighborhood. However, this testing strategy will help the residents of places like Bualda choose the safest tubewells for interim use until a treatment plant can be built.
All tubewells identified as safe by this 3-step process should be used as public drinking water supplies. These safe tubewells must be periodically monitored for As, Mn, U, Pb, Ni, Cr, B, Ba, and Mo. If a tubewell becomes unsafe, then an alternative drinking water supply must be identified, or the unsafe water must be treated.
Our earlier national-scale survey suggested that groundwater with unsafe levels of As, Mn, U, Pb, Ni, Cr, B, Ba, and Mo extend beyond Bangladesh’s borders into the 4 adjacent and densely populated Indian states of West Bengal, Assam, Meghalaya, and Tripura (Frisbie et al. 2002). The present neighborhood-scale survey in western Bangladesh borders the West Bengal districts of Nadia and 24-Parganas, where aquifers with similar characteristics occur (Bhattacharya et al. 2002). Thus, we urge that a similar survey be done in West Bengal to investigate possible exposure to unsafe levels of Mn, U, Pb, Ni, Cr, B, Ba and Mo in addition to As in drinking water.
 |
 |
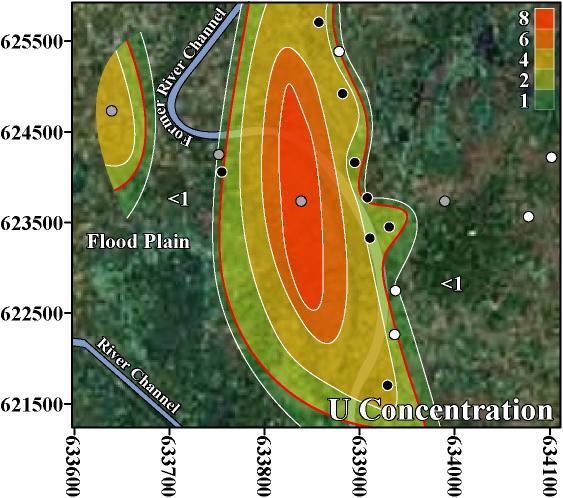 |
The inverse trend between As and U may be caused by the variability that is characteristic of delta-alluvial plain deposits from the Bengal Delta Plain in Bangladesh and West Bengal, India. For example, in Jamjami the concentration of As decreases with depth (p-value = 0.002, Figure 2), and the concentration of U increases with depth (p-value = 0.04, Figure 4). Komlapur, to some extent, also shows these trends. In contrast, Bualda and Fulbaria do not show any trends between As and depth, and U and depth. The aquifers in Jamjami and possibly Komlapur have medium-coarse grained sand at depth that was deposited in former river channels (Alam et al. 1990). The groundwater drawn into tubewells that are screened in these deposits may be under oxidizing conditions that remove As from groundwater and release U into groundwater. In contrast, the aquifers in all 4 neighborhoods have organic-rich mud at all depths that was deposited in flood plains (Alam et al. 1990). The groundwater drawn into tubewells that are screened in these deposits may be under reducing conditions that release As into groundwater and remove U from groundwater. Therefore, alluvial sediments of the Bengal Delta Plain make a complex 3-dimensional stratigraphy of medium-coarse grained sand and organic-rich mud deposits that may be responsible for the inverse trend between As and U. Other factors may also be controlling release of As and U. It is important to note that in areas where drilling deeper tubewells may access water with lower levels of As, the water from these deeper tubewells may contain increased levels of U, as was found in Jamjami and Komlapur.
Despite this inverse trend, 4% (3 out of 67) of the tubewells in this study had unsafe concentrations of both As and U. This is important because the home-scale drinking water filters that are being used in Bangladesh may not remove U. Also, up to 50% of Bangladesh’s tubewells exceed the WHO health-based drinking water guideline for U (BGS/DPHE 2001). The water treatment filters used in Bangladesh typically oxidize soluble As(III) to insoluble As(V) to remove As by absorption or precipitation. However, this oxidation may convert insoluble U(IV) to soluble U(VI) and potentially increase the U concentration of the water after treatment. Alternatively, this oxidation may keep dissolved U in the VI oxidation state and potentially cause no change in the U concentration of the water after treatment (Fairbridge 1972). Thus, these filters should be further evaluated for the removal of U.